Tyber Medical, LLC, a leading orthopaedic device manufacturer providing private label orthopaedic implants for the trauma, extremity, and spine markets, received a U.S. Food and Drug Administration (FDA) 501(k) clearance for its new line of foot and ankle plating systems. The FDA clearance covers over 42 different indication-specific anatomical plating families, extensively developed using a combination of CT scans, cadaveric labs, and consultation with surgical thought leaders.
Tyber Medical's plating systems are indicated to treat a comprehensive range of deformity, trauma, and degenerative conditions to the forefoot, midfoot, rearfoot, and ankle. Tyber Medical plans to launch the first phase of plates in the first half of 2021 and is actively pursuing the CE Mark in the second half of 2021.
Tyber Medical's Foot and Ankle Plating Systems feature a 30-degree, multi-angle, locking mechanism optimized for superior stability between the plate and screws. An intuitive, modular, tray delivery system is customizable with a colour-coded, clean layout that is consistent with the surgical flow to meet surgeons' instrumentation needs. The systems include low profile (1.3mm) plates to minimize soft tissue irritation while maximizing the variable locking technology to reduce screw head prominence. Additionally, the sterile and non-sterile plates include an engineered combination of material and surface modification which significantly increases fatigue resistance. "Our system is designed to minimize soft tissue trauma during the procedure," said Melissa Coale, Tyber Medical senior engineer.
"Our plates feature geometry to reduce overall prominence, especially in areas with minimal soft tissue coverage."
"We are excited to receive the FDA clearance that officially launches Tyber Medical into the orthopaedic plating arena," said Tyber Medical CEO and President, Jeff Tyber. "Our team has developed a robust method linking intrinsic design and ease of use for a variety of surgical indications. We expect to expand this methodology beyond the lower extremities and into all areas of the skeletal anatomy."
Tyber Medical's plating systems for fore-, mid-, hindfoot, ankle repair, and ankle fusion are indicated for use in the fixation of phalanges, metatarsals, and small bone fragments in the forefoot, and medium-to-large bones and multi-fragments in the mid- and hindfoot. Forty-two different indicated plates stabilize fractures, joint fusions, osteotomies, nonunions, malunions, reconstruction of small bones, revision surgeries, and re-plantations to facilitate successful healing in adult patients. The company's Ankle Fracture System is indicated for use in the fixation of fractures of the distal tibia, intra- and extra-articular, and medial malleolar fractures.
Tyber Medical is committed to developing rapid access, innovative, orthopaedic device technology to advance patient care and healing outcomes. The company's extensive medical device portfolio effectively supports gaps in its partners' product offerings in as little as four months though vertically-integrated design, engineering, manufacturing, quality management, regulatory, clinical research, and MDR compliance.
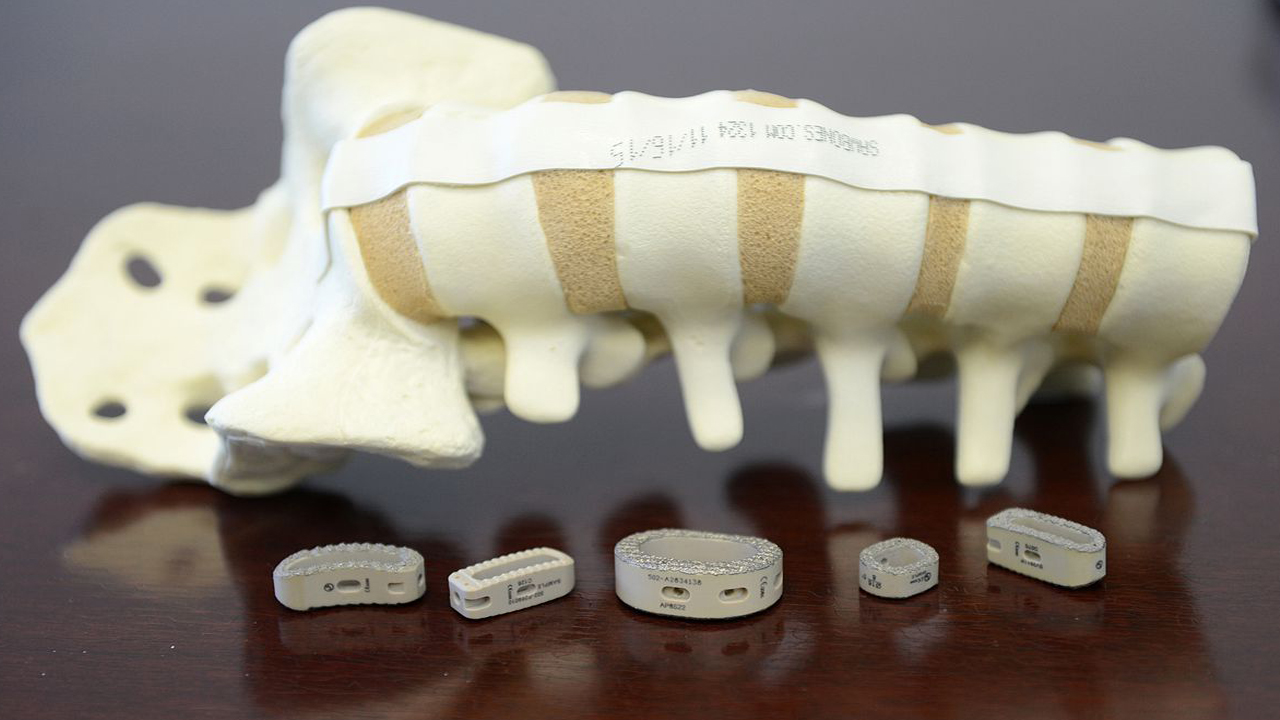
 The comprehensive portfolio is the first of several planned plating releases for the distal extremities
The comprehensive portfolio is the first of several planned plating releases for the distal extremities